Files
Download Full Text (479 KB)
Description
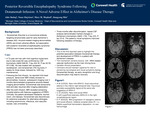
Background: Donanemab (Kisunla) is a monoclonal antibody targeting amyloid-beta used for early Alzheimer's disease (AD). Amyloid-related imaging abnormalities (ARIA) are known adverse effects, but association with posterior reversible encephalopathy syndrome (PRES) has not been previously described.
A 72-year-old right-handed man with mild OSA, IBS, asthma, and hyperlipidemia was followed for progressive memory loss since age 66, consistent with mild cognitive impairment due to late-onset Alzheimer's disease. CSF AD biomarkers (Aβ42 648.65, T-tau 424.35, P-tau 92.55, ATI 0.88) confirmed the diagnosis.
This case highlights a potential association between Donanemab therapy and development of PRES in a patient with Alzheimer's disease, possibly mediated by severe ARIA-related vascular dysfunction. Clinicians should maintain vigilance for new or worsening neurological and visual symptoms during Donanemab therapy, as early recognition and drug discontinuation may lead to recovery.
Publication Date
5-8-2026
Disciplines
Neurology
Recommended Citation
Shehaj A, HajAissa N, Mcphail Ciufo MM, Min J. Posterior reversible encephalopathy syndrome following donanemab infusion: A novel adverse effect in Alzheimer's disease therapy. Presented at: Research Day Corewell Health West; 2026 May 8; Grand Rapids, MI.

Comments
2026 Research Day Corewell Health West, Grand Rapids, MI, May 8, 2026. Abstract 2017